- About
-
Solutions
-
Services
- Biosciences
- Chemistry
- Integrated Drug Discovery
- Computer Aided Drug Design
- Hit Identification
- DMPK Services
- Target Classes and Modalities
- Therapeutic Areas
-
A-Z
- A
- B
- C
- D
- E
- F
- G
- H
- I
- K
- L
- M
- N
- O
- P
- R
- S
- T
- V
- X
-
Services
- Library
- News & Events
- Careers
GPCRs
Multi-disciplinary expertise to drive your GPCR programme forward
With our experience in GPCR cellular pharmacology, hit identification, protein purification, biophysics, and drug design, Domainex scientists are perfectly placed to support your GPCR projects at all stages of preclinical development.
G protein-coupled receptors (GPCRs) are one of the largest classes of membrane proteins and are targets for approximately 35% of clinically-available drugs.1 Despite the rise of other target classes and drug types, small molecule targeting of GPCRs remains at the forefront for the treatment of numerous pathologies. Domainex has extensive expertise in computational and medicinal chemistry, protein production, assay development, in vitro screening and molecular pharmacology for GPCRs. For an example of an integrated programme, see our poster on our collaboration with Parkinson’s UK in which we successfully identified lead-like negative allosteric modulators of the C5aR1.
Many GPCRs are located in the CNS and are targets for CNS diseases such as Parkinson’s and Alzheimer’s disease. See our separate CNS page to find out more about our expertise in CNS drug discovery.
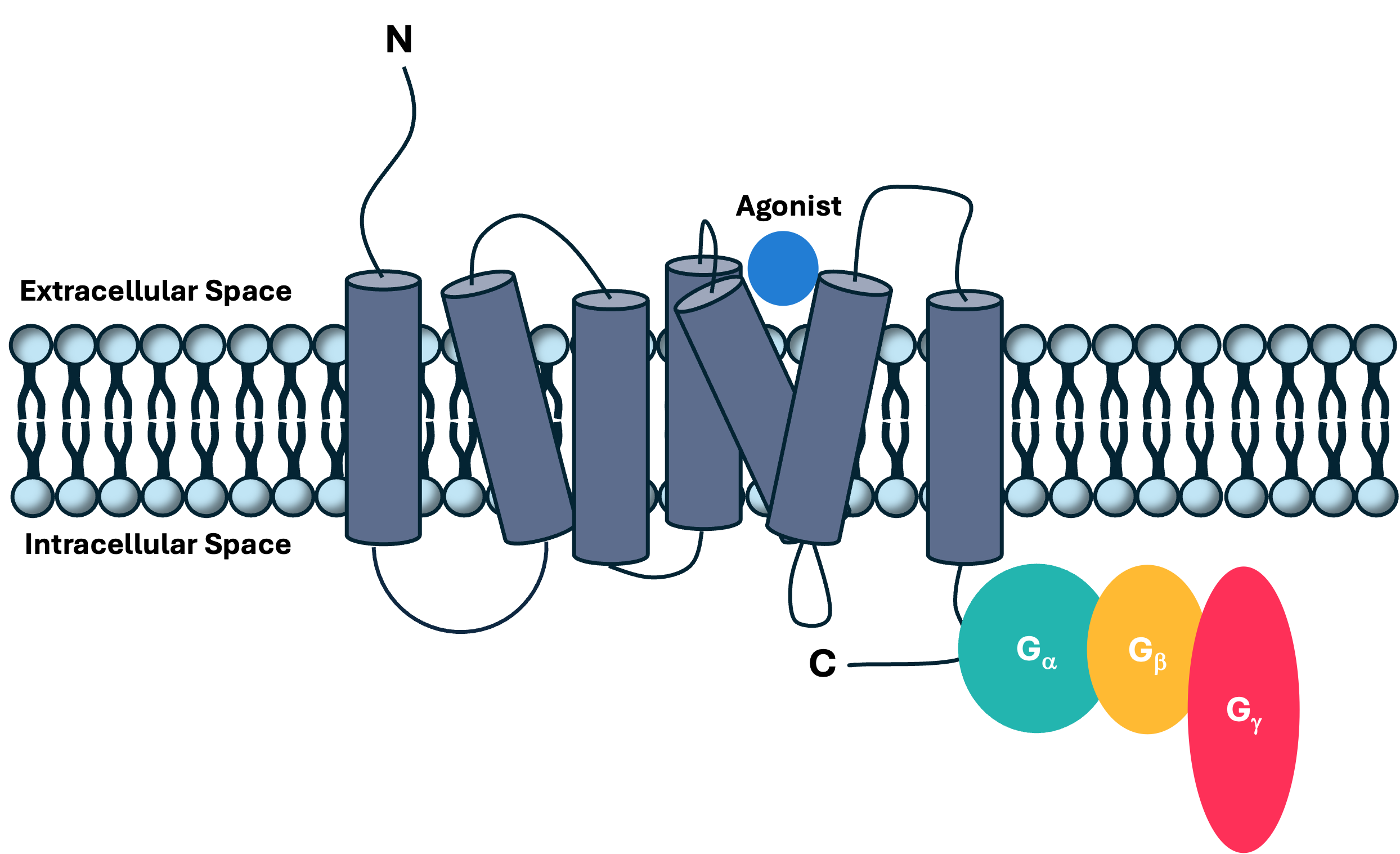
Figure 1: A schematic of a GPCR complex structure
Assay Development and Hit Identification
GPCRs signal via coupling to, and activating, hetero-trimeric G proteins (e.g. Gαi, Gαs and Gαq), which initiate diverse intracellular signalling cascades, often via multiple signalling pathways in response to a single ligand, driving ligand bias.
Depending on the project needs, Domainex can work with:
- Division-arrested cell lines expressing the GPCR of interest (for smaller scale project)
- Commercially available stable cell lines
- Stable cell lines generated in house (wild-type or tagged GPCR)
We leverage our in-house expertise of cellular biology and molecular pharmacology to develop bespoke assays to study most secondary messenger and intracellular signalling pathways and assess bias signalling and allostery.
- cAMP modulation
- Ca2+ mobilisation (FLIPR, Inositol Phosphate (IP1) accumulation)
- β-arrestin recruitment, internalisation and trafficking (imaging and flow cytometry)
- Downstream signalling (MAPK, chemotaxis, proliferation)
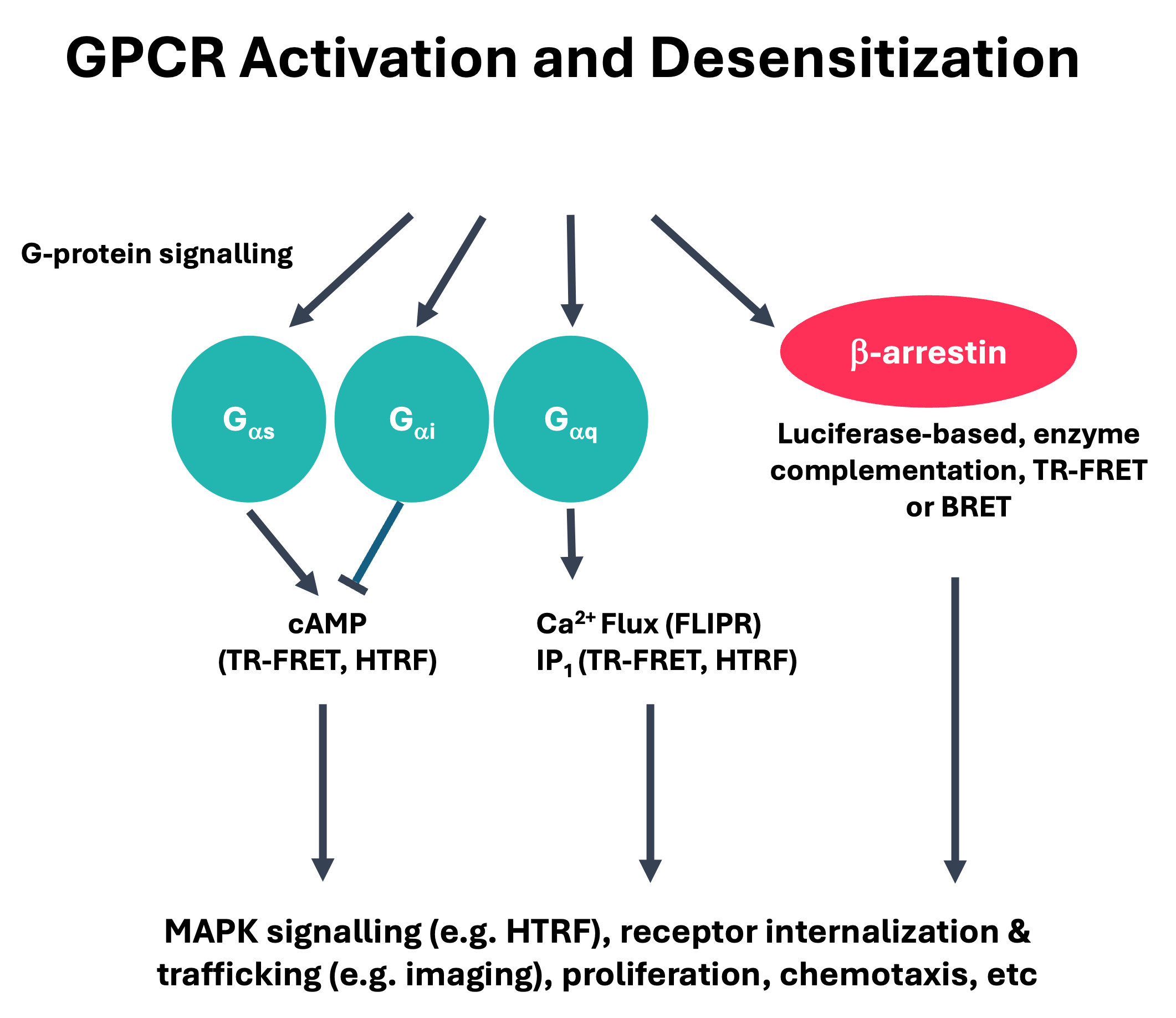
Figure 2: The assays systems that are typically developed for the various signalling pathways
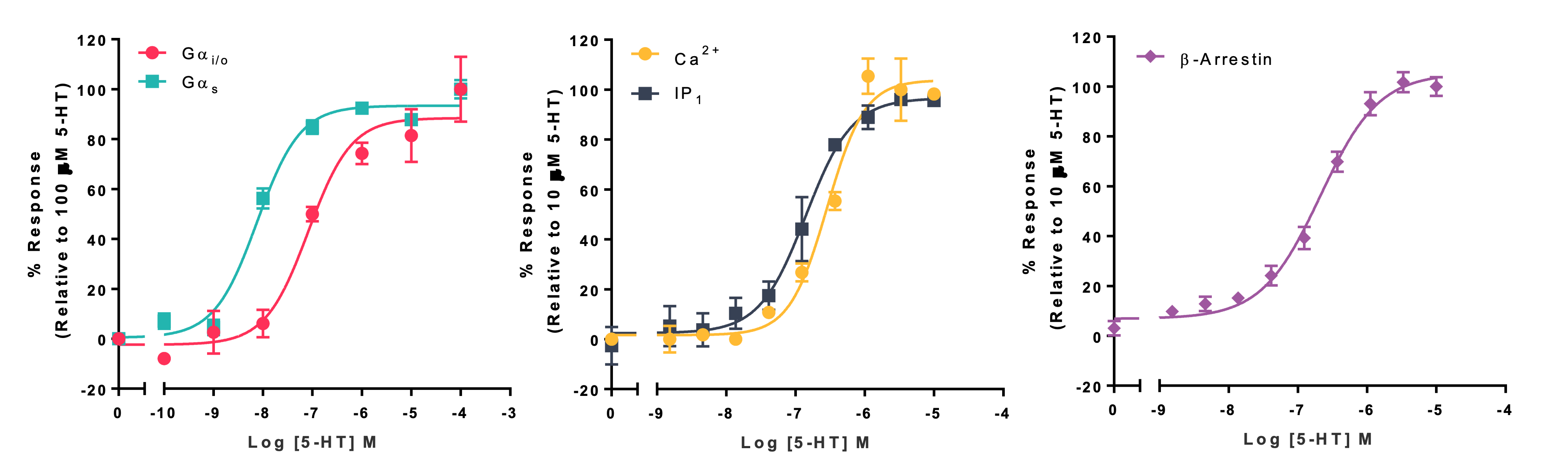
Figure 3: Example concentration response curve (CRC) data obtained upon 5-hydroxytryptamine (5-HT)-induced activation of various 5-hydroxytryptamine receptors (5-HTRs) expressed in CHO-K1 cells
Pharmacological Mechanism of Action analysis
Understanding detailed mechanism of action for GPCRs is key for progression through lead optimisation to help interpret structure-activity relationships (SAR), guide compound design and determine functional selectivity. Domainex regularly provides in-depth analysis for compounds acting via various mechanisms against GPCRs, including;
- Allosteric function (negative and positive allosteric modulators, NAMs, PAMs, Ago-NAM, Ago-PAM)
- Agonists (full and partial), antagonists, inverse agonists
- Binding or functional kinetics (on/off rate determination)
- pA2 analysis
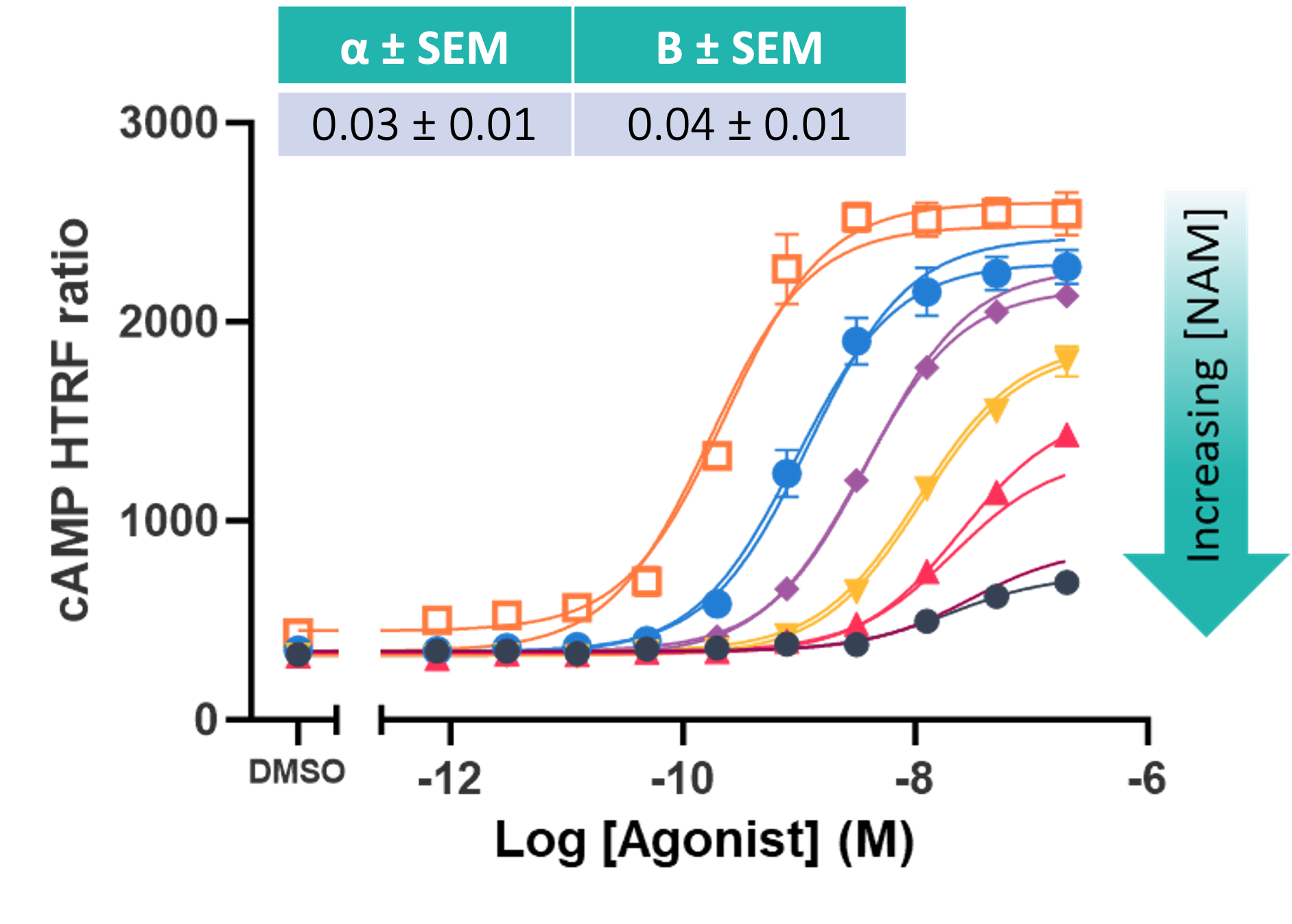
Figure 4: Example of a dose-shift assay for a negative allosteric modulator (NAM), quantifying α and β values to measure the effect of the compound modulating orthosteric affinity (KA) and efficacy (τA). Data is fitted with both a four-parameter logistic model and the operational model of allostery
GPCR Purification and Biophysics
Expressing recombinant, wild-type GPCRs and purifying them in their native confirmation has long been a technically demanding challenge. Domainex scientists have a breadth of experience with membrane protein expression, purification and biophysics. We work with our clients to provide a tailored solution for their target and application.
- Established membrane protein purification platform, optimised for GPCRs
- High quality QC for a range of downstream applications, e.g. structural biology of antibody generation
- Biophysical screening assays suitable for
- Hit ID screening (see our case study on fragment screening against the A2a receptor and Figure 5)
- Binding affinity determination for small molecules and biotherapeutics
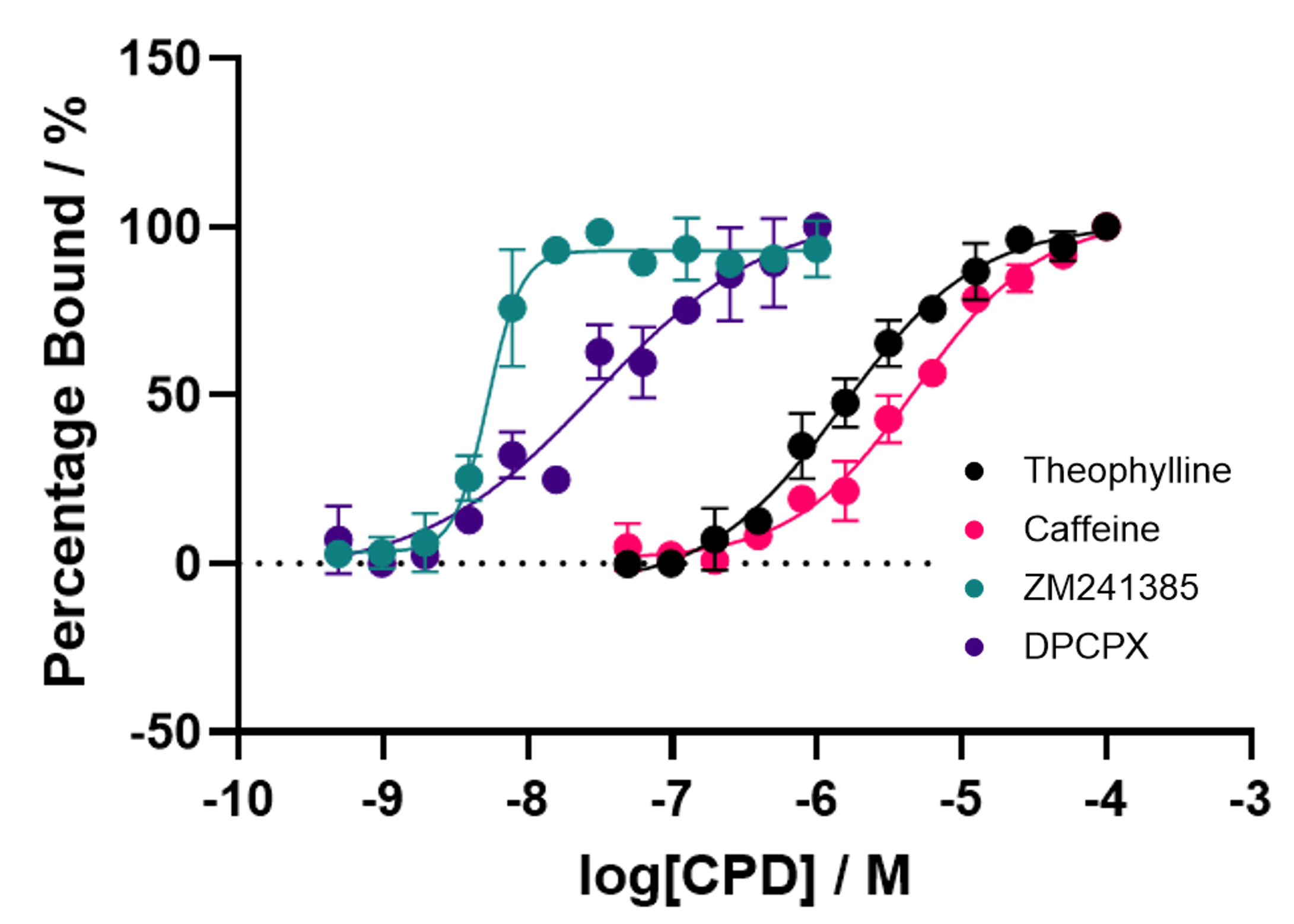
Figure 5: Spectral Shift assay affinity binding curves for four known A2aR small molecule ligands
Reference
- Sriram K, Insel PA. G Protein-Coupled Receptors as Targets for Approved Drugs: How Many Targets and How Many Drugs? Mol Pharmacol. 2018 Apr;93(4):251-258. doi: 10.1124/mol.117.111062. Epub 2018 Jan 3. PMID: 29298813; PMCID: PMC5820538.
Domainex was selected following an extensive review process on the basis of their experience and capability to execute a fully integrated drug discovery project, including the development of neuroinflammation assays and the optimisation of our compounds’ target engagement in the brain.
Dr Richard Morphy, Drug Discovery Manager at Parkinson’s UK
Frequently Asked Questions
Which GPCR Assay platforms and signalling pathways can Domainex work with?
Domainex supports a broad range of GPCR signalling modalities, covering both G protein–dependent and β arrestin–dependent pathways. Assay formats include:
- cAMP modulation assays using HTRF, AlphaScreen, or luminescence based detection for Gαs, Gαi/o pathways.
- IP₁ accumulation assays for Gαq coupled receptors using HTRF or ELISA based readouts.
- Calcium flux assays using fluorescent dyes or genetically encoded calcium indicators for high speed kinetic measurements.
- β arrestin recruitment assays using enzyme fragment complementation, BRET, or luminescence based biosensors.
- G protein activation assays including TRUPATH, NanoBiT based G protein dissociation, and BRET based G protein biosensors.
- Reporter gene assays for transcriptional outputs
Radioligand binding for affinity, selectivity, and kinetic characterisation - Internalization assays using flow cytometry
These platforms allow quantification of potency, efficacy, bias factors, and pathway selective pharmacology.
What GPCR pharmacology services do Domainex provide?
Domainex offers a full suite of GPCR discovery capabilities spanning early feasibility through lead optimisation:
- Functional assays for Gαs, Gαi/o, Gαq, and β‑arrestin pathways
- Radioligand binding (saturation, competition, kinetic)
- Custom assay development and optimisation
- High-throughput screening (HTS) and orthogonal confirmation assays
- Hit validation, mechanism‑of‑action studies, and bias quantification
- Selectivity profiling across GPCR families
- Data analysis, modelling, and SAR‑driven decision support
These services are designed to integrate seamlessly with medicinal chemistry and structural biology programmes
Domainex also have an established membrane purification platform, optimised for GPCRs, suitable for additional downstream applications such as biophysical assays.
What throughput levels are available?
Domainex supports:
- High throughput screening in 384 or 1536 well formats
- Medium throughput profiling for SAR cycles
- Low throughput mechanistic studies for detailed characterisation
Assay miniaturisation, robustness testing, and QX metrics (Z', S/B, CV%) ensure screening readiness
How does Domainex ensure data quality and reproducibility
Domainex quality control includes:
- Rigorous assay validation (Z’ factor, signal window, reproducibility)
- Reference agonist/antagonist benchmarking
- Replicate testing and plate‑uniformity studies
- Statistical QC and curve‑fit diagnostics
- Transparent reporting with raw data, fitted curves, and interpretation
Does Domainex offer GPCR selectivity panels?
Yes. Domainex can design custom selectivity panels across:
- Class A, B, and C GPCR families
- Therapeutic‑area‑focused panels (CNS, immunology, metabolic, cardiovascular)
- Binding or functional formats depending on receptor biology
Panels can be expanded to include ion channels, kinases, or phenotypic assays.
Which receptor expression systems can Domainex work with?
Domainex works with multiple expression systems to match biological relevance and assay robustness:
- Stable recombinant cell lines (CHO, HEK293, U2OS) for consistent expression and high‑throughput screening.
- Transient expression systems for rapid feasibility testing or low‑abundance receptors.
- Endogenous receptor systems to preserve native signalling context and accessory proteins.
- Membrane fractions for binding assays where whole‑cell signalling is not required.
For difficult‑to‑express GPCRs, Domainex can evaluate codon optimisation, signal peptides, chaperone co‑expression, or inducible expression systems.
Can Domainex work with biased agonism and mechanism of action studies for GPCRs?
Domainex supports quantitative analysis of ligand bias and mechanistic pharmacology:
- Determination of transduction coefficients across multiple pathways.
- Operational model fitting for partial agonists, antagonists, and allosteric modulators.
- Allosteric modulator characterisation including cooperativity factors (alpha, beta) and probe dependence.
- Inverse agonism and constitutive activity assessment.
- Kinetic pharmacology for on‑rate/off‑rate determination in functional assays.
What is the assay-development workflow for GPCR studies at Domainex?
Domainex follows a structured assay development workflow for cell-based assay optimisation:
- Selection of optimal cell line, receptor expression level, and detection modality.
- Determination of assay window, signal‑to‑background, Z’ factor, and EC80/IC50 reference conditions.
- Optimisation of buffer composition, incubation times, and plate format (96‑, 384‑, or 1536‑well).
- Validation of pharmacological standards to confirm expected potency and efficacy.
- Robustness testing for HTS readiness, including DMSO tolerance and plate‑uniformity studies.
What are the screening capabilities at Domainex for GPCRs?
Domainex can run multiple screening options:
- Primary HTS using miniaturised functional assays.
- Orthogonal assays to confirm mechanism and eliminate artefacts.
- Counter‑screens for off‑target GPCRs or non‑GPCR pathways.
- Profiling cascades to support SAR cycles with rapid turnaround.
Data are delivered with curve fits, QC metrics, and mechanistic interpretation.
How is data analysed and reported at Domainex for GPCR projects?
Domainex uses industry‑standard analysis pipelines:
- Non‑linear regression for EC50, IC50, Ki, and efficacy values.
- Bias quantification using Black–Leff, Ehlert, or Kenakin models.
- Kinetic modelling for association/dissociation and residence time.
- Statistical QC including replicate variability, plate‑level metrics, and outlier analysis.
Reports include raw data, fitted curves, assay validation metrics, and scientific interpretation.
What information is needed to initiate a project?
Typical requirements include:
- Target GPCR(s) and species
- Desired assay format(s) and readouts
- Compound numbers, formats, and handling requirements
- Project goals (screening, profiling, mechanistic studies, SAR support)
Domainex can advise on assay selection if the target strategy is still evolving. To speak with our experts please get in touch via our enquiries form.
Case studies
Start your next project with Domainex
Contact one of our experts today